EDITORS NOTE: I (Will) recently did a video on ARA which discusses a recent study that found ARA had positive effects on strength and muscle mass readers will want to check out. The results of this study will be covered in a future article by Monica when the study is published. This excellent article below by Monica discusses the long held belief this fatty acid is a negative for health and well being. As usual, the truth turns out to be more complex and a read of this article will cover the studies to demonstrate that. Part II of this article can be found HERE.
It is well known that the typical American diet provides too little omega-3 and too much omega-6, and thus has an elevated omega-6/omega-3 ratio. In turn, an elevated omega-6/omega-3 ratio has been linked to a number of common chronic diseases, notably cardiovascular diseases, inflammatory diseases, cancer, and certain psychiatric diseases such as depression.[1-3] The omega-6 fatty acid that has been vilified and blamed to give rise to these detrimental health outcomes is arachidonic acid (ARA).
However, a deeper look at the research data reveals that the association between omega-6 fats, especially ARA, with detrimental health outcomes isn’t as clear-cut as previously thought. Prominent lipid researchers are even questioning the value of the conceptually entrenched omega-6/omega-3 ratio. And there are indications that ARA, previously thought to be the omega-6 villain, actually may confer health benefits and even increase muscle mass and strength when combined with resistance training…in this 3-part series you will find out about it all…
Background to the health effects of omegs-6 and omega-3 essential fatty acids
ARA is the omega-6 fatty acid that competes with EPA (an omega-3 fatty acid) for incorporation into cell membranes. Once inside membranes, ARA and EPA compete for release (by phospholipases) and conversion into a myriad of potent substances called eicosanoids [4, 5], which play wide ranging roles in inflammation and regulation of immune function.[6, 7]
ARA has been deemed to be “bad” because it gives rise to pro-inflammatory eicosanoids.[8, 9] In contrast, the eicosanoids originating from EPA have different properties, and EPA and DHA also give rise to resolvins, which are anti-inflammatory and inflammation resolving.[9] EPA and DHA are therefore considered to be the “good” fatty acids.[8, 9]
Arguments to reduce intakes of ARA are based on the assumption that because ARA is a precursor for synthesis of a variety of pro-inflammatory eicosanoids, reducing ARA intakes should reduce cell membrane ARA content, which in turn should reduce generation of excessive inflammation and thereby lower risk of chronic disease (many of which have an inflammatory component in their development).[10]
In humans, supplementation with the long-chain omega-3 fatty acids EPA and DHA (fish oils) has a wide range of well-documented health benefits.[11-24] One of the most touted mechanism of action for the health promoting effects of fish oil has been indicated to be reduced levels of the omega-6 fatty acid ARA in cell membranes, and diminished production of ARA derived pro-inflammatory molecules.[5, 25, 26]
The “ARA is bad” theory is not consistently supported by human studies
However, this has not achieved consistent support in human studies.
INFLAMMATION
In a study of men and women aged 20-98 years, it was found that higher blood plasma levels of omega-6 fatty acids, mainly ARA, were associated with decreased plasma levels of pro-inflammatory markers and increased levels of anti-inflammatory markers.[27] Higher blood levels of both ARA and omega-3 fatty acids were associated with the lowest level of inflammatory markers.[27, 28] The combination of low ARA levels in cell membranes, together with low levels of EPA and DHA, causes perpetuation of the pro-inflammatory initiation phase and gives rise to chronic inflammation.[29] In line with this, in part 2 you will find out about ARA supplementation studies which show lack of impact on inflammatory markers.
DIABETES
Type 2 diabetics have reduced levels of ARA in blood [30] and platelet phospholipids [31] compared to non-diabetic healthy subjects. It is especially interesting that in diabetics, compared to non-diabetics, higher levels of glycosylated haemoglobin (HbA1c, a measure of long-term blood glucose levels) are associated with a significant reduction of ARA in platelet phospholipids, but not linoleic (LA, omega-6) or linolenic acid (ALA, omega-3).[31] Thus, one should expect an increase in platelet ARA (coupled with a decrease in platelet aggregation) after any intervention that improves glycaemic control in diabetes. [31]
CVD (cardiovascular disease)
Prospective studies of cardiovascular disease, which have measured blood or adipose tissue fatty acids in apparently healthy people with many years of follow up, have shown either no relation to omega-6 fatty acids or, more often, that low levels of ARA are actually predictive of a higher risk of cardiovascular disease and cancer.[32-39]
A Japanese study compared blood fatty acid levels and cardiovascular disease mortality in residents of fisher and farmer villages.[40, 41] It was found that the residents in the fisher villages had higher levels of all three main long-chain fatty acids, ARA +EPA + DHA [40], and exhibited the lowest cardiovascular disease mortality.[41]
The most recent systematic review and meta-analysis of the association of fatty acids and heart disease found that blood ARA levels was associated with a reduced risk for heart disease.[42] In one study of middle-aged men initially free of coronary heart disease at baseline, it was found that those who developed a fatal or non-fatal heart attack (myocardial infarction) or died suddenly during a follow-up of 5-7 years had lower ARA and EPA levels in blood phospholipids at baseline, compared with those who did not develop heart disease.[35]
Another study analyzed the fatty acid composition of phospholipids in coronary (heart) arteries in 30 cases of sudden cardiac death due to heart disease (aged 40 +/- 5 years) and in 29 traffic accident victims (aged 45 +/- 6 years).[37] As expected, the coronary arteries from cases of sudden cardiac death had more atherosclerotic lesions than those of controls. However, unexpectedly, levels of LA was significantly higher and the levels of ARA and of all the other major polyunsaturated fatty acids (both n-6 and n-3) was significantly lower in cases of sudden cardiac death than in controls.[37] This is in line with evidence of a trend toward increased CVD risk associated with higher LA blood levels and lower ARA levels.[39]
Associations between the two major n-6 fatty acids LA and ARA, and blood cholesterol lipoprotein sub-classes, were examined in middle-age men (40-49 years of age). [43] It was found that ARA was significantly associated with a more beneficial blood cholesterol lipoprotein distribution (less atherogenic LDL and VLDL particles and a more protective HD particle profile) than LA.[43]
ARA is also inversely associated with plasminogen activator inhibitor-1 (PAI-1) in healthy-middle aged men (but not in older men aged 70 years[44]).[45] PAI-1 has an anti-fibrinolytic function (i.e. is stimulates blood clot formation) [46], and high levels of PAI-1 are associated with an increased risk for developing heart disease and/or stroke [47-51] and cancer.[49, 51] An inverse association means that higher blood levels of ARA are associated with lower levels of PAI-1.
OSTEOPOROSIS
Men with the highest intakes of ARA had an 80% lower risk of hip fracture than those in the lowest intakes after a 6 year long follow-up, even after adjustment for total fat intake and baseline bone mineral density.[52]
CANCER
A recent large systematic review of observational studies concluded that intake and tissue levels of ARA are not associated with increased breast or prostate cancer risk.[53]
The importance of distinguishing the different omega-6 fatty acids LA and ARA
The term omega-6 is commonly used to refer to both LA and ARA, and it is often assumed that LA that is consumed will easily get converted to ARA in the body.[25, 54, 55] However, detailed tracer studies with stable isotopes have shown that the conversion of LA to ARA is only 0.2-0.6%.[56, 57] Other studies have confirmed that LA intake is not associated with elevations in ARA in tissue membranes.[58, 59]
These data clearly demonstrate the poor rate of conversion of LA to AA in the body, even in healthy adults consuming large amounts of LA. This was confirmed in a systematic reviewed of the human literature on changes in LA intake and its subsequent impact on changing tissue ARA in erythrocytes and plasma/serum phospholipids.[60] Decreasing LA intake by up to 90% was not significantly correlated with changes in ARA levels in the phospholipid pool of plasma/serum.[60] Similarly, when LA Intakes were increased up to six fold, no significant correlations with ARA levels were observed. [60] Only intake of ARA was found to increase ARA levels in plasma/serum phospholipids.[60] It is notable that several studies consistently found that ARA supplementation does not affect cell membrane phospholipid levels of EPA or DHA .[61-64]
High intake of LA may inhibit incorporation of EPA from fish oil into cell membranes and thereby increases the ARA/EPA ratio (without affecting ARA levels) [58, 59, 65], while a low intake of LA has the opposite effect.[59] Although an intake of 10.5% energy from LA with a dietary LA:ALA ratio of 10:1 (which is typical in the standard American diet) results in a lower plasma phospholipid levels of EPA, it does not increase levels of ARA compared with an intake of 3.8% energy from LA with a LA:ALA ratio of 4:1.[59] Studies actually show that increased levels of LA in red blood cell membranes [66] and blood plasma phospholipids [58] are associated with lower levels of not only EPA and DHA, but also ARA.
The inefficient conversion of LA to ARA, coupled with the disparate effects and associations of LA and ARA with health outcomes (as outlined above), indicates that discussions about omega-6 fatty acids have to clearly state which omega-6 fatty acid is being referred to. It also shows that the concept of an optimal ratio between omega-6/omega-3 fats in the diet can be very misleading.
The bell-shaped relationship between ARA and EPA + DHA in cell membranes
It is commonly believed that increasing intakes of EPA and DHA (eg. fish oil) will reduce levels of ARA in cell membranes.[67, 68] However, the relationship between ARA and EPA + DHA is not linear. Several studies have found that ARA levels may rise, rather than fall, in response to EPA and DHA supplementation in people who have sub-optimal levels of EPA and/or DHA.[69] For example, in omega-3 deficient subjects, treatment with EPA resulted in a striking elevation in red blood cell ARA content.[69] Other human studies which have looked at ARA levels during supplementation of various types of EPA/DHA preparations have shown that phospholipid ARA levels rise initially when the total dose of EPA and DHA administered is relatively small.[70-73] The commonly reported fall in ARA levels only comes later when the cumulative amount of EPA and DHA administered is high, or with prolonged fish oil supplementation.
This is because the relation between the omega-3 index and arachidonic acid is bell-shaped, synergistic at low EPA+DHA status and antagonistic at high EPA+DHA status, as illustrated in the graph (click on the graph to see full image).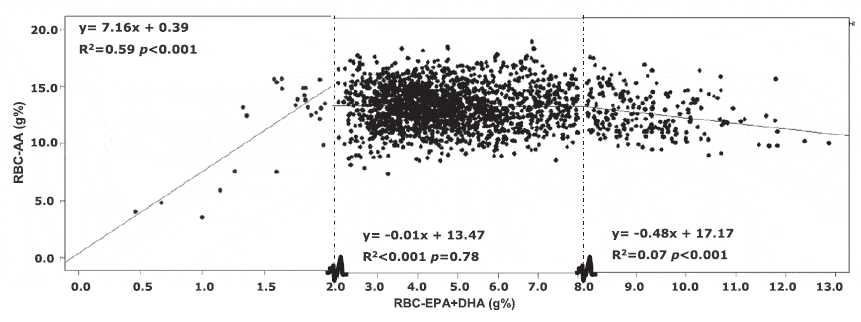
Graph showing the relationship between EPA+DHA and AA levels in red blood cells (RBC). Cut-off levels are at RBC-EPA+DHA below 2 g% for a synergistic relation and at RBC-EPA+DHA over 8 g% for an antagonistic relationship between RBC-EPA+DHA and RBC-AA. Note the bell-shape (inverted U-shape) of the curve.
Thus, if you are taking a high-dose fish oil, you may actually be deficient in ARA. The best way to find out is to do an omega-3 index blood test, which measures the fatty acid composition in red blood cells. The omega-3 index is a validated surrogate for omega-3 fatty acid status. [74-76] Some omega-3 blood tests, like the Omega Score offered by Life Extension, also provide a complete breakdown of fatty acids by weight, including the ARA/EPA ratio and the omega-6/omega-3 ratio. Knowing your ARA/EPA ratio is especially relevant if you decide to try supplementing with ARA. Doing a before-after omega-3 index and ARA/EPA ratio test when supplementing with ARA is strongly recommended; that way you can relate changes in your fatty acid status to possible muscle, strength performance enhancements (more on that in part 3) and find your own optimal ARA and EPA+DHA level.
Coming next in part 2…
Now that you know that ARA is not the bad guy as previously thought, the next logical question is if ARA supplementation can possibly confer beneficial effects and if supplementing with ARA is safe. I will tackle that in part 2. Stay tuned…
PART II HERE
References:
1. Simopoulos, A.P., A. Leaf, and N. Salem, Jr., Workshop statement on the essentiality of and recommended dietary intakes for Omega-6 and Omega-3 fatty acids. Prostaglandins Leukot Essent Fatty Acids, 2000. 63(3): p. 119-21.
2. Okuyama, H., T. Kobayashi, and S. Watanabe, Dietary fatty acids–the N-6/N-3 balance and chronic elderly diseases. Excess linoleic acid and relative N-3 deficiency syndrome seen in Japan. Prog Lipid Res, 1996. 35(4): p. 409-57.
3. Lands, W.E., Commentary on the workshop statement. Essentiality of and recommended dietary intakes for Omega-6 and Omega-3 fatty acids. Prostaglandins Leukot Essent Fatty Acids, 2000. 63(3): p. 125-6.
4. Harris, W.S., The omega-6/omega-3 ratio and cardiovascular disease risk: uses and abuses. Curr Atheroscler Rep, 2006. 8(6): p. 453-9.
5. Wada, M., et al., Enzymes and receptors of prostaglandin pathways with arachidonic acid-derived versus eicosapentaenoic acid-derived substrates and products. J Biol Chem, 2007. 282(31): p. 22254-66.
6. Lewis, R.A., K.F. Austen, and R.J. Soberman, Leukotrienes and other products of the 5-lipoxygenase pathway. Biochemistry and relation to pathobiology in human diseases. N Engl J Med, 1990. 323(10): p. 645-55.
7. Tilley, S.L., T.M. Coffman, and B.H. Koller, Mixed messages: modulation of inflammation and immune responses by prostaglandins and thromboxanes. J Clin Invest, 2001. 108(1): p. 15-23.
8. Calder, P.C., n-3 fatty acids, inflammation and immunity: new mechanisms to explain old actions. Proc Nutr Soc, 2013. 72(3): p. 326-36.
9. Calder, P.C., Fatty acids and inflammation: the cutting edge between food and pharma. Eur J Pharmacol, 2011. 668 Suppl 1: p. S50-8.
10. Harris, W.S., et al., Omega-6 fatty acids and risk for cardiovascular disease: a science advisory from the American Heart Association Nutrition Subcommittee of the Council on Nutrition, Physical Activity, and Metabolism; Council on Cardiovascular Nursing; and Council on Epidemiology and Prevention. Circulation, 2009. 119(6): p. 902-7.
11. Adkins, Y. and D.S. Kelley, Mechanisms underlying the cardioprotective effects of omega-3 polyunsaturated fatty acids. J Nutr Biochem, 2010. 21(9): p. 781-92.
12. Chang, C.L. and R.J. Deckelbaum, Omega-3 fatty acids: mechanisms underlying ‘protective effects’ in atherosclerosis. Curr Opin Lipidol, 2013. 24(4): p. 345-50.
13. Calder, P.C., The role of marine omega-3 (n-3) fatty acids in inflammatory processes, atherosclerosis and plaque stability. Mol Nutr Food Res, 2012. 56(7): p. 1073-80.
14. Swanson, D., R. Block, and S.A. Mousa, Omega-3 fatty acids EPA and DHA: health benefits throughout life. Adv Nutr, 2012. 3(1): p. 1-7.
15. Casula, M., et al., Long-term effect of high dose omega-3 fatty acid supplementation for secondary prevention of cardiovascular outcomes: A meta-analysis of randomized, double blind, placebo controlled trials. Atheroscler Suppl, 2013. 14(2): p. 243-51.
16. Crupi, R., A. Marino, and S. Cuzzocrea, n-3 fatty acids: role in neurogenesis and neuroplasticity. Curr Med Chem, 2013. 20(24): p. 2953-63.
17. Mozaffarian, D. and J.H. Wu, Omega-3 fatty acids and cardiovascular disease: effects on risk factors, molecular pathways, and clinical events. J Am Coll Cardiol, 2011. 58(20): p. 2047-67.
18. Peter, S., S. Chopra, and J.J. Jacob, A fish a day, keeps the cardiologist away! – A review of the effect of omega-3 fatty acids in the cardiovascular system. Indian J Endocrinol Metab, 2013. 17(3): p. 422-9.
19. Merendino, N., et al., Dietary omega -3 polyunsaturated fatty acid DHA: a potential adjuvant in the treatment of cancer. Biomed Res Int, 2013. 2013: p. 310186.
20. Calder, P.C., Omega-3 fatty acids and inflammatory processes. Nutrients, 2010. 2(3): p. 355-74.
21. Lorente-Cebrian, S., et al., Role of omega-3 fatty acids in obesity, metabolic syndrome, and cardiovascular diseases: a review of the evidence. J Physiol Biochem, 2013. 69(3): p. 633-51.
22. Xin, W., W. Wei, and X. Li, Effects of fish oil supplementation on cardiac function in chronic heart failure: a meta-analysis of randomised controlled trials. Heart, 2012. 98(22): p. 1620-5.
23. Balakumar, P. and G. Taneja, Fish oil and vascular endothelial protection: bench to bedside. Free Radic Biol Med, 2012. 53(2): p. 271-9.
24. Kromhout, D., et al., Fish oil and omega-3 fatty acids in cardiovascular disease: do they really work? Eur Heart J, 2012. 33(4): p. 436-43.
25. Fritsche, K.L., Too much linoleic acid promotes inflammation-doesn’t it? Prostaglandins Leukot Essent Fatty Acids, 2008. 79(3-5): p. 173-5.
26. Schmitz, G. and J. Ecker, The opposing effects of n-3 and n-6 fatty acids. Prog Lipid Res, 2008. 47(2): p. 147-55.
27. Ferrucci, L., et al., Relationship of plasma polyunsaturated fatty acids to circulating inflammatory markers. J Clin Endocrinol Metab, 2006. 91(2): p. 439-46.
28. Pischon, T., et al., Habitual dietary intake of n-3 and n-6 fatty acids in relation to inflammatory markers among US men and women. Circulation, 2003. 108(2): p. 155-60.
29. Bosma-den Boer, M.M., M.L. van Wetten, and L. Pruimboom, Chronic inflammatory diseases are stimulated by current lifestyle: how diet, stress levels and medication prevent our body from recovering. Nutr Metab (Lond), 2012. 9(1): p. 32.
30. Suhre, K., et al., Metabolic footprint of diabetes: a multiplatform metabolomics study in an epidemiological setting. PLoS One, 2010. 5(11): p. e13953.
31. Jones, D.B., et al., Low phospholipid arachidonic acid values in diabetic platelets. Br Med J (Clin Res Ed), 1983. 286(6360): p. 173-5.
32. Horrobin, D.F., Abnormal membrane concentrations of 20 and 22-carbon essential fatty acids: a common link between risk factors and coronary and peripheral vascular disease? Prostaglandins Leukot Essent Fatty Acids, 1995. 53(6): p. 385-96.
33. Siguel, E.N. and R.H. Lerman, Altered fatty acid metabolism in patients with angiographically documented coronary artery disease. Metabolism, 1994. 43(8): p. 982-93.
34. Wood, D.A., et al., Adipose tissue and platelet fatty acids and coronary heart disease in Scottish men. Lancet, 1984. 2(8395): p. 117-21.
35. Miettinen, T.A., et al., Fatty-acid composition of serum lipids predicts myocardial infarction. Br Med J (Clin Res Ed), 1982. 285(6347): p. 993-6.
36. Kingsbury, K.J., et al., A comparison of the polyunsaturated fatty acids of the plasma cholesteryl esters and subcutaneous depot fats of atheromatous and normal people. Clin Sci, 1962. 22: p. 161-70.
37. Luostarinen, R., M. Boberg, and T. Saldeen, Fatty acid composition in total phospholipids of human coronary arteries in sudden cardiac death. Atherosclerosis, 1993. 99(2): p. 187-93.
38. de Goede, J., et al., N-6 and N-3 fatty acid cholesteryl esters in relation to fatal CHD in a Dutch adult population: a nested case-control study and meta-analysis. PLoS One, 2013. 8(5): p. e59408.
39. de Oliveira Otto, M.C., et al., Circulating and dietary omega-3 and omega-6 polyunsaturated fatty acids and incidence of CVD in the Multi-Ethnic Study of Atherosclerosis. J Am Heart Assoc, 2013. 2(6): p. e000506.
40. Hirai, A., et al., Eicosapentaenoic acid and platelet function in Japanese. Lancet, 1980. 2(8204): p. 1132-3.
41. Hirai, A., [An epidemiological study on dietary ingestion of eicosapentaenoic acid (EPA) and platelet function in Japanese]. Nihon Naika Gakkai Zasshi, 1985. 74(1): p. 13-20.
42. Chowdhury, R., et al., Association of Dietary, Circulating, and Supplement Fatty Acids With Coronary Risk: A Systematic Review and Meta-analysis. Annals of Internal Medicine. 2014 Mar;160(6):398-406, 2014.
43. Choo, J., et al., Serum n-6 fatty acids and lipoprotein subclasses in middle-aged men: the population-based cross-sectional ERA-JUMP study. Am J Clin Nutr, 2010. 91(5): p. 1195-203.
44. Byberg, L., et al., Plasminogen activator inhibitor-1 and relations to fatty acid composition in the diet and in serum cholesterol esters. Arterioscler Thromb Vasc Biol, 2001. 21(12): p. 2086-92.
45. Lee, S., et al., Significant inverse associations of serum n-6 fatty acids with plasma plasminogen activator inhibitor-1. Br J Nutr, 2012. 107(4): p. 567-72.
46. Ha, H., E.Y. Oh, and H.B. Lee, The role of plasminogen activator inhibitor 1 in renal and cardiovascular diseases. Nat Rev Nephrol, 2009. 5(4): p. 203-11.
47. Senno, S.L. and L. Pechet, Clinical implications of elevated PAI-1 revisited: multiple arterial thrombosis in a patient with essential thrombocythemia and elevated plasminogen activator inhibitor-1 (PAI-1) levels: a case report and review of the literature. J Thromb Thrombolysis, 1999. 8(2): p. 105-12.
48. Thogersen, A.M., et al., High plasminogen activator inhibitor and tissue plasminogen activator levels in plasma precede a first acute myocardial infarction in both men and women: evidence for the fibrinolytic system as an independent primary risk factor. Circulation, 1998. 98(21): p. 2241-7.
49. Donati, M., et al., An atypical presentation of intrahepatic perforated cholecystitis: a modern indication to open cholecystectomy. Report of a case. BMC Surg, 2014. 14: p. 6.
50. Fortenberry, Y.M., Plasminogen activator inhibitor-1 inhibitors: a patent review (2006-present). Expert Opin Ther Pat, 2013. 23(7): p. 801-15.
51. Gramling, M.W. and F.C. Church, Plasminogen activator inhibitor-1 is an aggregate response factor with pleiotropic effects on cell signaling in vascular disease and the tumor microenvironment. Thromb Res, 2010. 125(5): p. 377-81.
52. Farina, E.K., et al., Dietary intakes of arachidonic acid and alpha-linolenic acid are associated with reduced risk of hip fracture in older adults. J Nutr, 2011. 141(6): p. 1146-53.
53. Sakai, M., et al., Arachidonic acid and cancer risk: a systematic review of observational studies. BMC Cancer, 2012. 12: p. 606.
54. Wall, R., et al., Fatty acids from fish: the anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr Rev, 2010. 68(5): p. 280-9.
55. Laye, S., Polyunsaturated fatty acids, neuroinflammation and well being. Prostaglandins Leukot Essent Fatty Acids, 2010. 82(4-6): p. 295-303.
56. Demmelmair, H., et al., Comparison of bolus versus fractionated oral applications of [13C]-linoleic acid in humans. Eur J Clin Invest, 1999. 29(7): p. 603-9.
57. Hussein, N., et al., Long-chain conversion of [13C]linoleic acid and alpha-linolenic acid in response to marked changes in their dietary intake in men. J Lipid Res, 2005. 46(2): p. 269-80.
58. Angela Liou, Y. and S.M. Innis, Dietary linoleic acid has no effect on arachidonic acid, but increases n-6 eicosadienoic acid, and lowers dihomo-gamma-linolenic and eicosapentaenoic acid in plasma of adult men. Prostaglandins Leukot Essent Fatty Acids, 2009. 80(4): p. 201-6.
59. Liou, Y.A., et al., Decreasing linoleic acid with constant alpha-linolenic acid in dietary fats increases (n-3) eicosapentaenoic acid in plasma phospholipids in healthy men. J Nutr, 2007. 137(4): p. 945-52.
60. Rett, B.S. and J. Whelan, Increasing dietary linoleic acid does not increase tissue arachidonic acid content in adults consuming Western-type diets: a systematic review. Nutr Metab (Lond), 2011. 8: p. 36.
61. Kusumoto, A., et al., Effects of arachidonate-enriched triacylglycerol supplementation on serum fatty acids and platelet aggregation in healthy male subjects with a fish diet. Br J Nutr, 2007. 98(3): p. 626-35.
62. Nelson, G.J., et al., The effect of dietary arachidonic acid on platelet function, platelet fatty acid composition, and blood coagulation in humans. Lipids, 1997. 32(4): p. 421-5.
63. Hirota, S., et al., Low-dose arachidonic acid intake increases erythrocytes and plasma arachidonic acid in young women. Prostaglandins Leukot Essent Fatty Acids, 2010. 83(2): p. 83-8.
64. Kakutani, S., et al., Supplementation of arachidonic acid-enriched oil increases arachidonic acid contents in plasma phospholipids, but does not increase their metabolites and clinical parameters in Japanese healthy elderly individuals: a randomized controlled study. Lipids Health Dis, 2011. 10: p. 241.
65. Cleland, L.G., et al., Linoleate inhibits EPA incorporation from dietary fish-oil supplements in human subjects. Am J Clin Nutr, 1992. 55(2): p. 395-9.
66. Friesen, R.W. and S.M. Innis, Linoleic acid is associated with lower long-chain n-6 and n-3 fatty acids in red blood cell lipids of Canadian pregnant women. Am J Clin Nutr, 2010. 91(1): p. 23-31.
67. Horrobin, D.F., et al., Eicosapentaenoic acid and arachidonic acid: collaboration and not antagonism is the key to biological understanding. Prostaglandins Leukot Essent Fatty Acids, 2002. 66(1): p. 83-90.
68. Martinelli, N., L. Consoli, and O. Olivieri, A ‘desaturase hypothesis’ for atherosclerosis: Janus-faced enzymes in omega-6 and omega-3 polyunsaturated fatty acid metabolism. J Nutrigenet Nutrigenomics, 2009. 2(3): p. 129-39.
69. Bjerve, K.S., et al., alpha-Linolenic acid and long-chain omega-3 fatty acid supplementation in three patients with omega-3 fatty acid deficiency: effect on lymphocyte function, plasma and red cell lipids, and prostanoid formation. Am J Clin Nutr, 1989. 49(2): p. 290-300.
70. Hansen, J.B., et al., Comparative effects of prolonged intake of highly purified fish oils as ethyl ester or triglyceride on lipids, haemostasis and platelet function in normolipaemic men. Eur J Clin Nutr, 1993. 47(7): p. 497-507.
71. von Schacky, C., et al., The effect of dietary omega-3 fatty acids on coronary atherosclerosis. A randomized, double-blind, placebo-controlled trial. Ann Intern Med, 1999. 130(7): p. 554-62.
72. Reis, G.J., et al., Effects of two types of fish oil supplements on serum lipids and plasma phospholipid fatty acids in coronary artery disease. Am J Cardiol, 1990. 66(17): p. 1171-5.
73. Zuijdgeest-van Leeuwen, S.D., et al., Incorporation and washout of orally administered n-3 fatty acid ethyl esters in different plasma lipid fractions. Br J Nutr, 1999. 82(6): p. 481-8.
74. Harris, W.S., Omega-3 fatty acids and cardiovascular disease: a case for omega-3 index as a new risk factor. Pharmacol Res, 2007. 55(3): p. 217-23.
75. Harris, W.S. and C. Von Schacky, The Omega-3 Index: a new risk factor for death from coronary heart disease? Prev Med, 2004. 39(1): p. 212-20.
76. von Schacky, C., Omega-3 index and cardiovascular health. Nutrients, 2014. 6(2): p. 799-814.






Excellent article. Maybe the culprit was LA all along. Eggs have lots of ARA. Surely the gestation-birth process would involve good things not bad. I may look into the Omega test of LEF/Labcorp to see where I am at. This is also further proof that one can not rely on the past ans always has to be willing to review the past in a critical light, to see it it still measures up. Best ARA article I have ever seen yet. Lots of careful distinctions made in study and its interpretation, too. I give it 5 stars!
Thank you. Parts 2 and 3 are even better! 😉
It’s not just the best article of it’s kind on the topic, it’s actually a ground breaking article that will cause a shift in paradigm regarding ARA, LA, and their interplay to EPA/DHA. And as Monica said, Part II and III, even better! 🙂
I’m assuming Will still recommends the one-third rule in the e-book? (1/3 fats from polyunsaturated fats, 1/3 monounsaturated, 1/3 saturated)
If you get your fats from unprocessed foods, as long as you get your essential fatty acids, the exact proportion of saturated, mono- and polyunsaturated fat is not as important.
Also, against the background of a reduced carb diet, the body becomes efficient in metabolizing saturated fat for energy, and there is no need to restrict saturated fat intake.
Such a great article! I’ve been looking into omega 3’s and omega 6’s and have found that many people who have both mental and physical diseases are low in arachidonic acid. I was wondering if that means their diet was low in it, making them more vulnerable to the sickness, or is it that the inflammatory process is breaking it down in their bodies. Like in Alzheimer’s-would more dietary AA plus EPA, as in chicken broth and eggs from pastured chickens, with are high in arachidonic acid, be helpful to restore AA in these people?